PIPELINE
ALA-1000 : Long-acting release injection for treating opioid use disorder

Rationale for Drug Development: The treatment for opioid addiction is a chronic process. Currently, the medication-assisted treatment (MAT) drugs are the mainstream of treating substance use disorders. However, the bottlenecks of traditional oral MAT drugs, such as missing doses and potential of drug abuse and misuse, have no longer meet the medical need of patients. The development of long-acting products can effectively improve the shortcomings of oral products. ALA-1000 has distinct characteristics as compared with the competitive products including low viscosity, small injection volume, and smooth drug release profile, which result in fewer side effects and improve drug adherence and medical compliance, providing a better medical choice for patients.
In-Relar Long-Acting Release Platform :
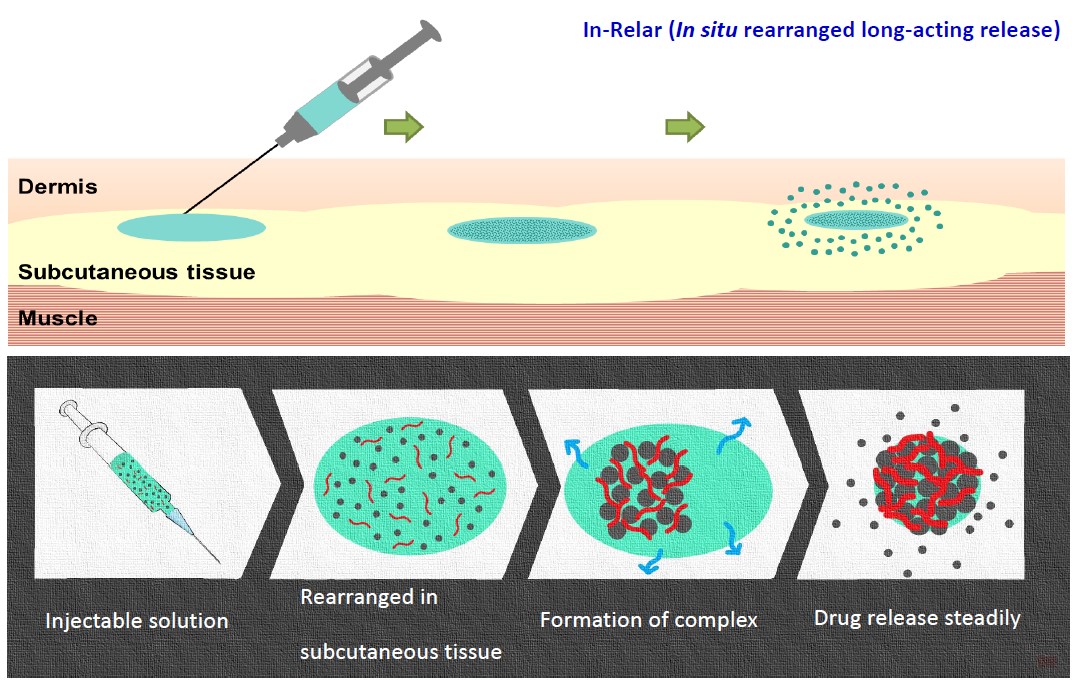
In-Relar platform: It is a novel drug delivery platform developed by Alar. After subcutaneous injection, the drug solidifies to form a depot. The active pharmaceutical ingredient will then be released steadily from the depot, maintaining a drug level above the therapeutic concentration for several weeks to months.
